Free EU-Wide Shipping on Orders Over €200
This product is intended solely for use as a research chemical in vitro and laboratory experimentation by licensed, qualified professionals.
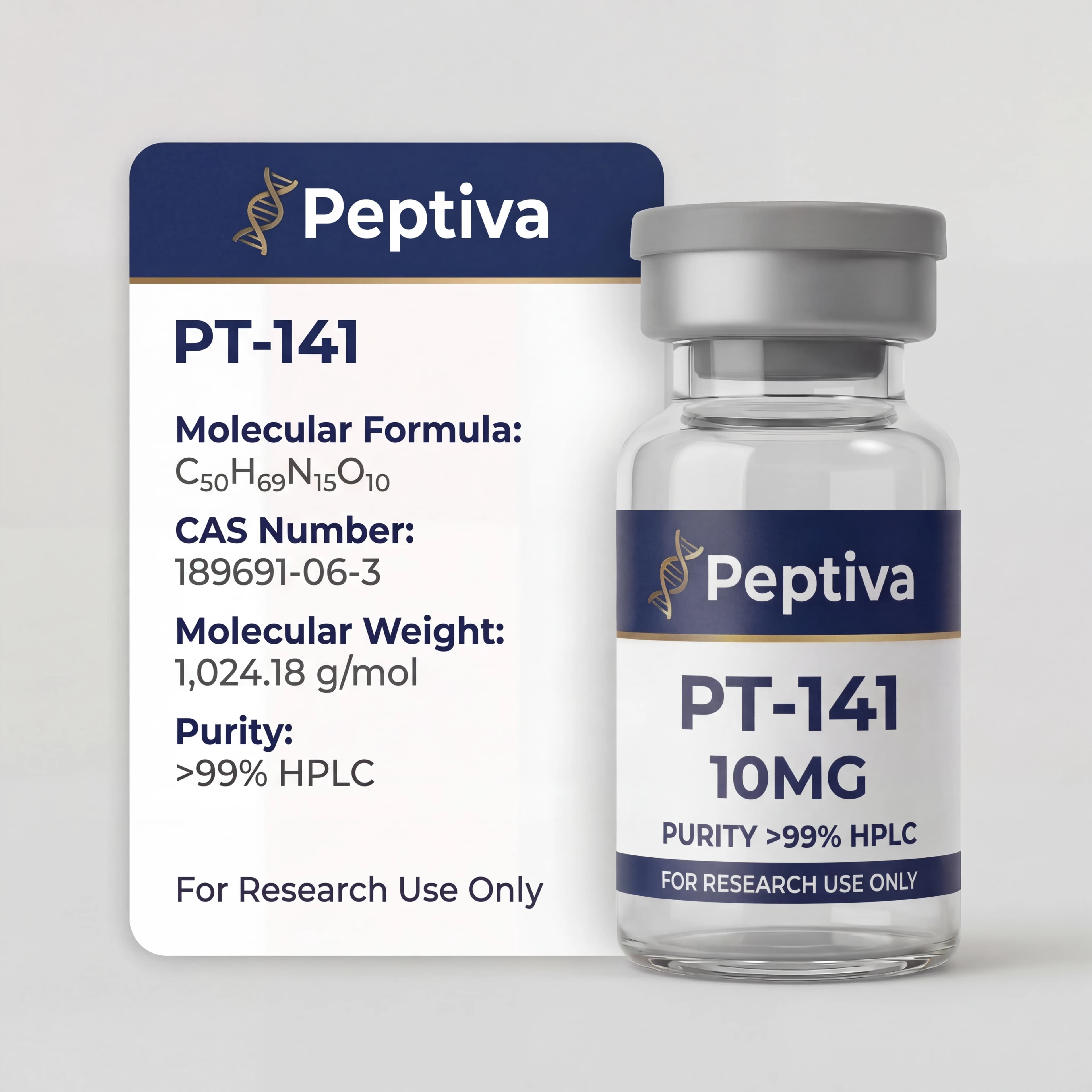
PT-141
40,00 €
PT-141 is a synthetic melanocortin receptor agonist derived from Melanotan II. Researched for its effects on melanocortin pathways involved in sexual function and behavior. 10mg lyophilized powder, ≥99% purity.
Bulk Discounts
- Buy 3+ for 38,00 € each and save 5%
- Buy 5+ for 36,00 € each and save 10%
- Buy 10+ for 34,00 € each and save 15%
Ships Tomorrow - order cutoff passed
Free EU Shipping on orders over €200
Third party tested
PT-141—
40,00 €
Advanced Laboratory Testing
HPLC
Mass Spec
≥99% Purity
Secure
Fast Ship
EU Based
Characteristics
| Molecular Formula | C50H68N14O10 |
| CAS | 189691-06-3 |
| Molar Mass | 1025.18 g/mol |
| Amino Acid Sequence | Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-OH |
| Synonyms | Bremelanotide, PT141 |
| Solubility | Soluble in water |
| Organoleptic Profile | White to off-white powder |
| Physical Form | Lyophilized powder |
How does PT-141 work?
PT-141 activates melanocortin receptors (primarily MC3R and MC4R) in the hypothalamus. Unlike PDE5 inhibitors that act on vascular mechanisms, PT-141 works through the central nervous system.
The peptide's CNS-mediated effects make it useful for research into the neural pathways underlying sexual motivation and arousal responses. These receptors are involved in diverse physiological processes including energy homeostasis, inflammation, and sexual behavior.
PT-141 helps researchers understand the structure-activity relationships of melanocortin peptides and the development of receptor-selective agonists.
Benefits
- Melanocortin Research: Pharmacological tool for investigating MC3R and MC4R receptor function and signaling
- Behavioral Neuroscience: Research into neural pathways underlying sexual motivation and arousal responses
- Receptor Pharmacology: Aids understanding of structure-activity relationships of melanocortin peptides
- CNS Mechanism: Unique central nervous system mechanism distinct from vascular-based approaches
Side Effects
PT-141 is generally considered safe and well tolerated when used in research settings. However, as with any substance, there is a potential for side effects, particularly when used at high concentrations or for prolonged periods.
- Nausea: Most commonly reported effect, typically transient and dose-dependent
- Flushing: Facial flushing reported in study subjects
- Headache: Mild headache may occur during initial administration
- Blood Pressure Changes: Transient increases in blood pressure observed in some studies
It's important to note that the safety and efficacy of PT-141 for human use have not been fully established, and more research is needed to determine the long-term effects of this peptide complex.
Summary
PT-141 (Bremelanotide) is a synthetic cyclic heptapeptide and melanocortin receptor agonist. Originally developed as a derivative of Melanotan II during tanning peptide research, its effects on sexual function were discovered incidentally.
The FDA approved bremelanotide (Vyleesi) in 2019 for hypoactive sexual desire disorder in premenopausal women, validating the melanocortin pathway as a therapeutic target.
Certificate of Analysis (COA) available upon request. Each batch is tested via HPLC for purity and Mass Spectrometry for identity confirmation.
References
- 1Kingsberg SA, et al. "Bremelanotide for the Treatment of Hypoactive Sexual Desire Disorder" Obstetrics & Gynecology. 2019. doi:10.1097/AOG.0000000000003500
- 2Clayton AH, et al. "Bremelanotide for female sexual dysfunctions in premenopausal women: a randomized, placebo-controlled dose-finding trial" Women's Health. 2016. doi:10.2217/whe-2016-0018
Resource
All products on this site are for research and development use only. Products are not for human consumption of any kind. The statements made on this website have not been evaluated by the US Food and Drug Administration or the European Medicines Agency. Products are not intended to diagnose, treat, cure, or prevent any disease.