Free EU-Wide Shipping on Orders Over €200
This product is intended solely for use as a research chemical in vitro and laboratory experimentation by licensed, qualified professionals.
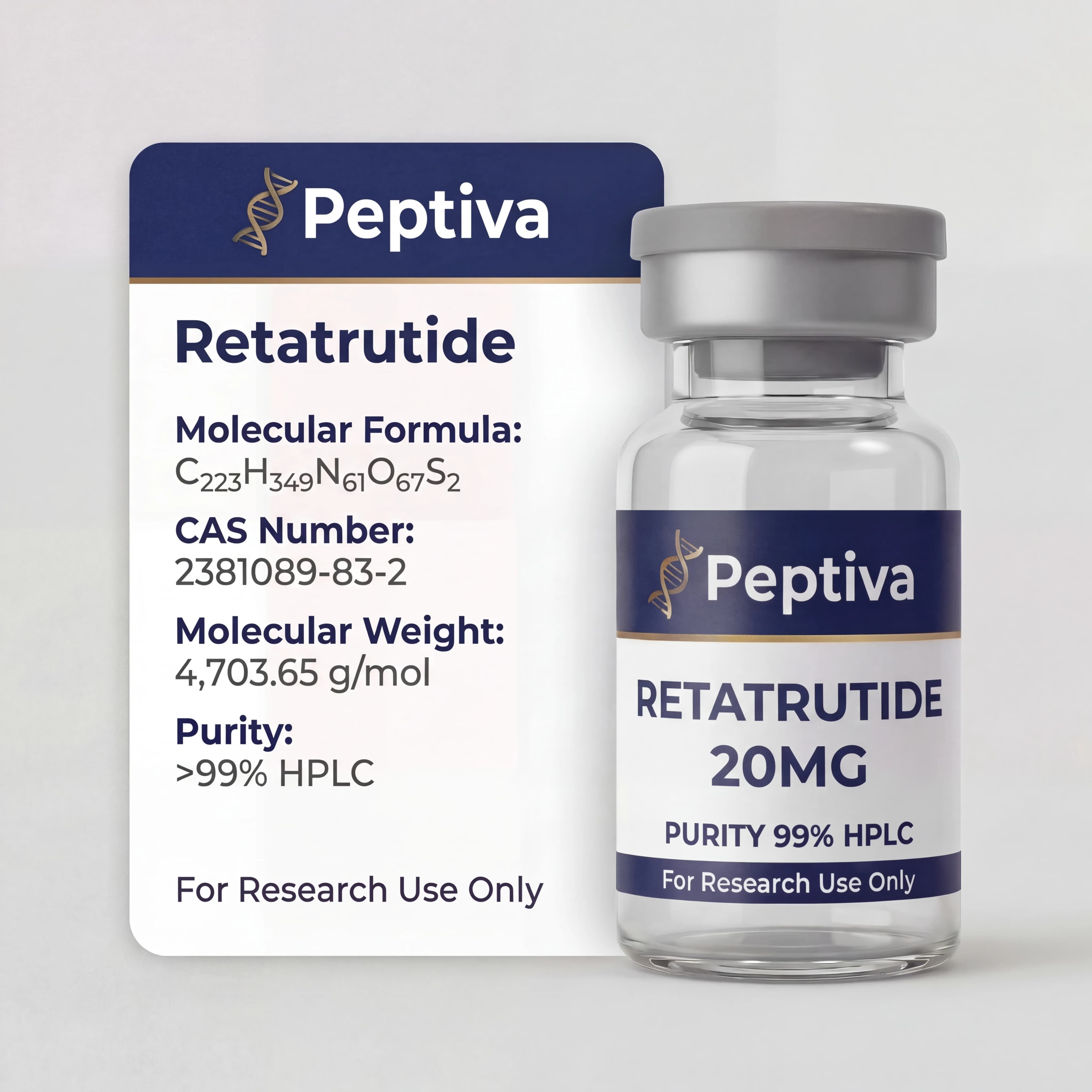
Retatrutide
130,00 €
Retatrutide is a novel triple-agonist peptide targeting GIP, GLP-1, and glucagon receptors simultaneously. Investigated for metabolic research applications including glucose homeostasis and energy expenditure. 20mg lyophilized powder, ≥99% purity.
Bulk Discounts
- Buy 3+ for 123,50 € each and save 5%
- Buy 5+ for 117,00 € each and save 10%
- Buy 10+ for 110,50 € each and save 15%
Ships Tomorrow - order cutoff passed
Free EU Shipping on orders over €200
Third party tested
Retatrutide—
130,00 €
Advanced Laboratory Testing
HPLC
Mass Spec
≥99% Purity
Secure
Fast Ship
EU Based
Characteristics
| Molecular Formula | C205H322N56O63S2 |
| CAS | 2381089-83-2 |
| Molar Mass | ~4605.5 g/mol |
| Synonyms | LY3437943, Eli Lilly Triple Agonist |
| Solubility | Soluble in water |
| Organoleptic Profile | White to off-white powder |
| Physical Form | Lyophilized powder |
How does Retatrutide work?
Retatrutide is a triple hormone receptor agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique multi-receptor mechanism offers potentially synergistic effects.
The GLP-1 component enhances insulin sensitivity and promotes satiety. The GIP component further augments insulin secretion and may influence fat metabolism. The glucagon receptor activation is of particular interest for its potential thermogenic effects and influence on hepatic glucose output.
Unlike single- or dual-agonist compounds, the triple mechanism allows for modulation of multiple metabolic pathways simultaneously, making Retatrutide valuable for research into metabolic regulation.
Benefits
- Metabolic Research: Significant effects on glucose homeostasis and insulin sensitivity observed in clinical trials
- Energy Expenditure: Glucagon component may influence hepatic glucose output and energy expenditure
- Body Composition: Improved body composition metrics observed across multiple dose levels
- Triple Receptor Activity: Unique simultaneous activation of GIP, GLP-1, and glucagon receptors
- Appetite Regulation: Promotes satiety and reduces food intake through central and peripheral mechanisms
Side Effects
Retatrutide is generally considered safe and well tolerated when used in research settings. However, as with any substance, there is a potential for side effects, particularly when used at high concentrations or for prolonged periods.
- Gastrointestinal Effects: Nausea and vomiting are the most commonly reported effects, particularly during dose escalation
- Injection Site Reactions: Mild reactions at the injection site may occur
- Fatigue: Temporary fatigue during the initial adjustment period
- Hypoglycemia Risk: Potential risk of low blood sugar in certain conditions, particularly with concomitant insulin use
It's important to note that the safety and efficacy of Retatrutide for human use have not been fully established, and more research is needed to determine the long-term effects of this peptide complex.
Summary
Retatrutide (LY3437943) represents a new class of investigational peptide developed by Eli Lilly. As a triple hormone receptor agonist, it has generated significant research interest due to its unique multi-receptor mechanism.
Phase 2 clinical trials demonstrated dose-dependent effects on metabolic parameters with a manageable safety profile. The compound is currently being evaluated in Phase 3 studies.
Certificate of Analysis (COA) available upon request. Each batch is tested via HPLC for purity and Mass Spectrometry for identity confirmation.
References
- 1Jastreboff AM, et al. "Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial" New England Journal of Medicine. 2023. doi:10.1056/NEJMoa2301972
- 2Rosenstock J, et al. "Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-comparator-controlled, parallel-group, phase 2 trial" The Lancet. 2023. doi:10.1016/S0140-6736(23)01053-X
Resource
All products on this site are for research and development use only. Products are not for human consumption of any kind. The statements made on this website have not been evaluated by the US Food and Drug Administration or the European Medicines Agency. Products are not intended to diagnose, treat, cure, or prevent any disease.